Differential Aeration Corrosion takes place when there is an uneven supply of oxygen to areas of the same metal component. It is a type of electrochemical corrosion that affects metals such as steel and iron. When a poorly oxygenated area is adjacent to an area with a good supply of oxygen, an anodic/cathodic reaction occurs. Oxygen screening, crevice corrosion and poultice action are other terms for this type of corrosion.
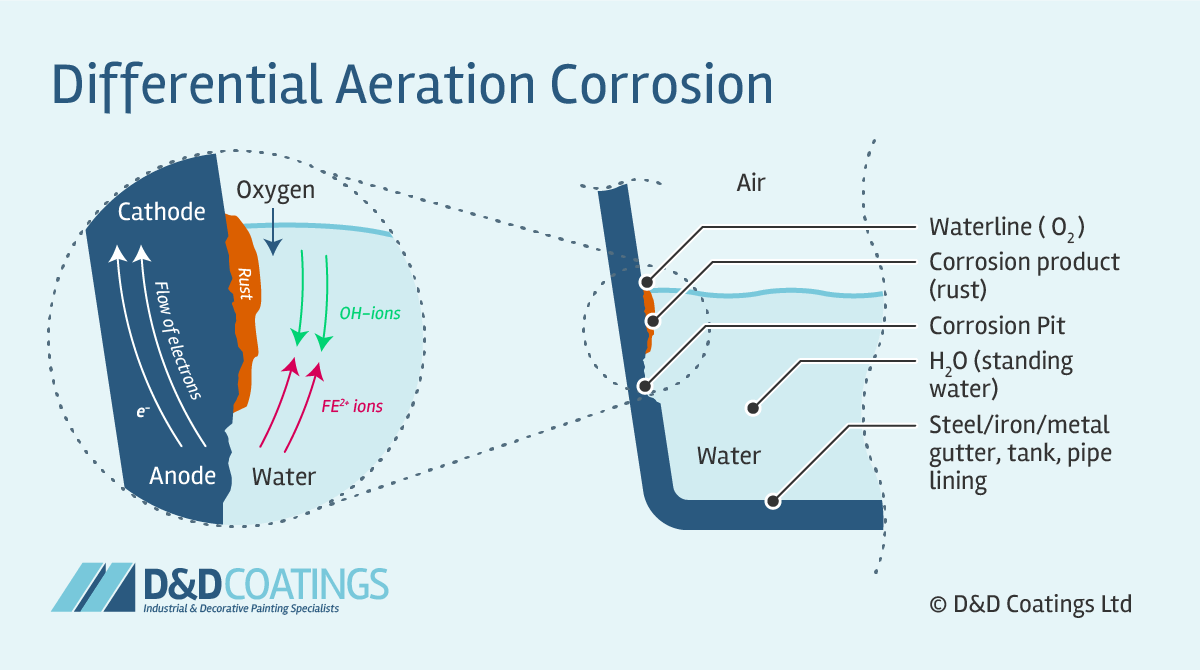
The reaction occurs because oppositely charged electrons flow between the smaller anode and larger cathode. Positively charged cations meeting negatively charged anions forming corrosion product and a resulting pit in the metal, otherwise known as pitting corrosion. In a gutter, pipe, tank or similar the anode is just below the waterline. This is where the oxidation occurs, corrosion product forms and a pit develops weakening the metal.
In the diagram below you can see how the cathode action reduces oxygen from the air to produce hydroxide ions. When the ions between the cathodic and anodic areas meet iron hydroxide forms, precipitates and oxidises to form the corrosive material or rust. The diagram shows an instance of waterline corrosion. The equation is O2 + 2H2O + 4e– → 4OH– (cathode) and Fe2+ + 2e– → Fe– (anode).
Where does differential aeration corrosion occur?
Differential aeration corrosion often happens in steel guttering that isn’t sufficiently protected or kept clean and clear from blockage. Standing water in a gutter system combined with a coating that isn’t adequately protecting the metal will result in the formation of corrosion along the waterline. The same applies to metal tanks and pipes. If water or a corrosive liquid is allowed to stand where there is a good supply of oxygen, the electrochemical reaction will occur.
Other instances would be when a water droplet settles on exposed steel. The area below the water droplet is low on oxygen while the surrounding area has plenty of oxygen from the air. This creates a cathodic and anodic reaction. The cathodic reaction depletes the oxygen beneath the water droplet. This area is now anodic and starts to corrode forming a pit.
What is waterline corrosion?
Waterline corrosion occurs when differential aeration is present in a neutral medium. It is usually found in steel guttering, pipes and tanks that are poorly maintained or damaged. The protected cathodic part is above the waterline and the area starved of oxygen is just below, which is where corrosion develops.
Summary
Read more about differential aeration corrosion here. If you would like to find out about our corrosion treatment services please call 01606 554040 or visit our contact page.



